Featured Remediation Approach
Tersus Advanced Surface Kinetics (TASK™) liberates NAPL and captures them with enhanced recovery techniques.
Surfactant-Enhanced Aquifer Remediation
Tersus provides surfactant formulations specific to site geochemistry and NAPL composition. Our TASK™ chemistry is based on a fundamental understanding of microemulsions and capillary phenomena.
The force that prevents NAPL from flowing inside an aquifer is called the capillary force. This force arises because of the high energy surface that exists between oil and water. Because physical systems move to the lowest energy environments (thermodynamics), the oil and water seek to minimize the contact between them. The NAPL cannot flow because flowing involves increasing the area of contact.
Surfactants can help increase the rate of removing NAPL from an aquifer by two different mechanisms. One mechanism is called solubilization. Here, surfactant micelles provide little mobile pools of oil that can dissolve some of the NAPL, so it can be removed within the flowing groundwater. This will work but requires that the volume of surfactant exceeds the volume of NAPL, which can be very expensive.
The other way that surfactants help remove NAPL is to lower the energy of the area of contact between the oil and water. The interfacial tension between the oil and water is direct measure of the energy of the interface. When surfactants adsorb at the oil/water interface, the energy of the interface decreases. When the energy of the interface decreases, the barrier that prevents the NAPL from flowing in the subsurface also decreases and more of the NAPL can be pumped directly to the surface as a separate non-aqueous phase liquid (NAPL).
Because there is a direct connection between oil/water interfacial tension and mobilization of subsurface NAPL, Tersus focuses its formulation efforts on minimizing the interfacial tension between the oil and water. Therefore, we always request samples of site groundwater, site NAPL, and site soil before finalizing a site-specific surfactant formulation. Laboratory testing with site materials results in a surfactant system which produces the lowest possible oil/water interfacial tension.
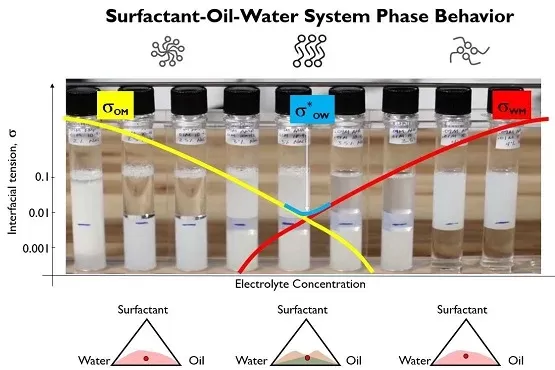
Now as it happens, the phase behavior of systems containing oil, water and surfactants is reliable guide to finding the lowest possible interfacial tension. This always occurs in the presence of what we call a middle phase microemulsion, a separate thermodynamic phase that contains roughly equal amounts of oil and water, stabilized by layers of surfactants. When the surfactant system is a little too water soluble, the surfactant pulls the oil into the water and forms a swollen water phase that we call an oil-in-water microemulsion. When the surfactant is a little too oil soluble the surfactant pulls water into the oil phase and forms a water-in-oil microemulsion. However, when the surfactant is balanced in its affinity for the NAPL and the groundwater, it forms the middle phase microemulsion. The lowest possible NAPL/groundwater interfacial tension is always found under the conditions where the middle phase microemulsions forms; consequently, the maximum possible mobilization of aquifer NAPL will occur with a surfactant formulation that produces a middle phase microemulsion. When we receive site NAPL and groundwater, it uses those materials to assure that the surfactant formulation shipped to the site always produces a middle phase microemulsion.
While minimizing oil/water interfacial tension is critical to maximizing NAPL mobilization and minimizing the amount of surfactant required, it also has another significant benefit: it simplifies the treatment and disposal of the produced oil and water. We often can design systems that use 0.9% by weight surfactant.
Most people know surfactants as additives that are used to stabilize oil/water emulsions; but not all surfactant formulations will produce stable oil/water emulsions. In fact, the most stable oil/water emulsions are formed by surfactant systems that produce oil-in-water microemulsions, where the surfactant is a little too water soluble. Emulsion stability decreases if the oil/water interfacial tension becomes too low and the oil drops no longer have a stable, spherical shape. This is exactly what happens in the middle phase microemulsion systems. The interfacial tension becomes too low for the oil drops to have a stable shape; thus, an emulsion made from oil and water in the presence of a middle phase surfactant system will break spontaneously. This eliminates the need to add chemical additives or use off-site disposal of a large volume of NAPL contaminated groundwater.
Consequently, Tersus first develops an effective middle phase surfactant formulation to create an ultra-low interfacial tension between the site NAPL and the site groundwater, and then we tweak the formulation to produce a fast-breaking emulsion.
Because of our carefully designed laboratory testing and formulation process, Tersus ships to your site a surfactant formulation that will simultaneously produce an ultra-low NAPL/groundwater interfacial tension, thereby maximizing the mobilization of NAPL in the aquifer, while also producing only fast breaking NAPL/groundwater emulsions that can be broken easily on the surface, reducing off site waste disposal and increasing the opportunity to recycle the recovered NAPL.